Metal-Organic Frameworks: MOFs
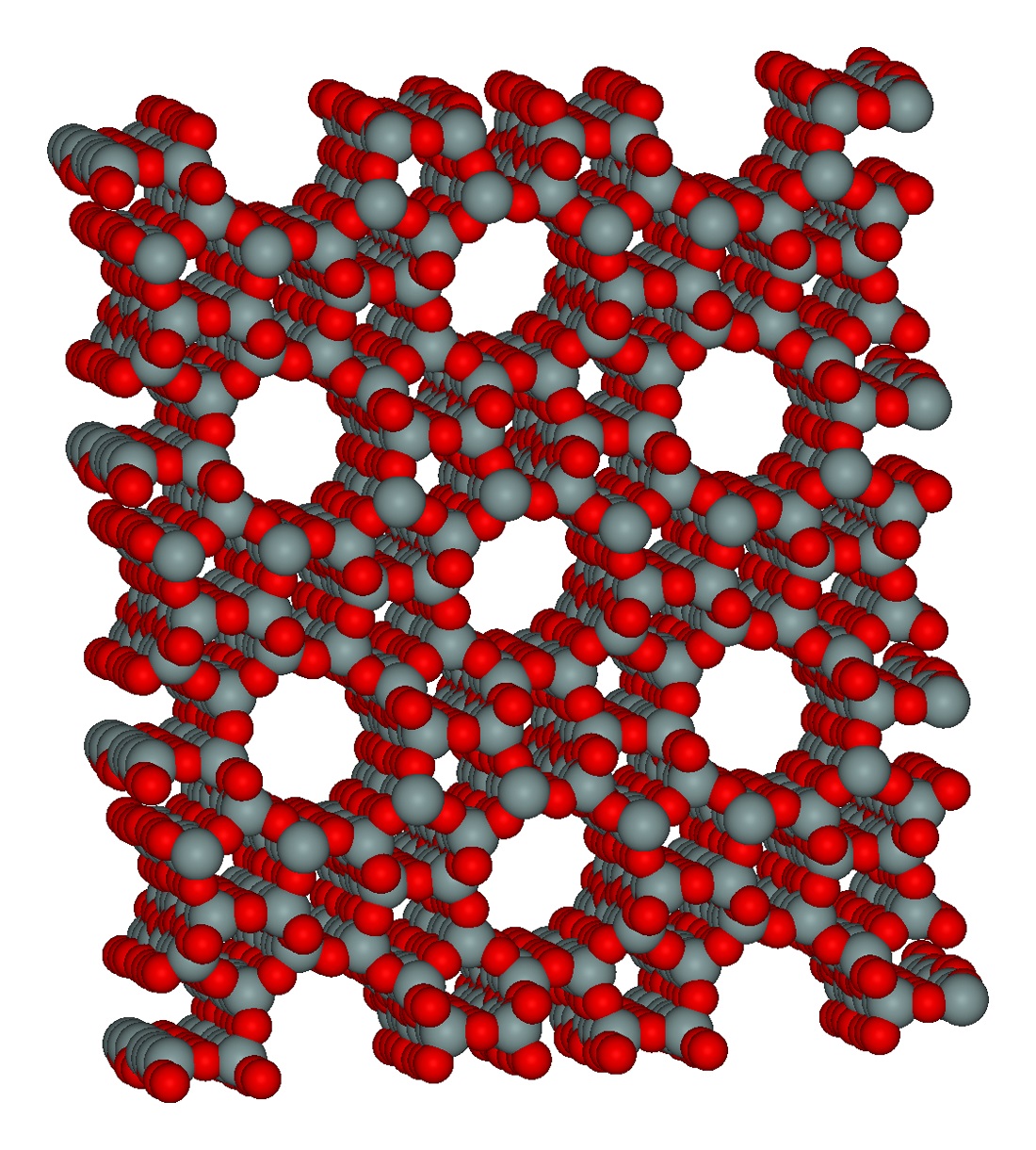
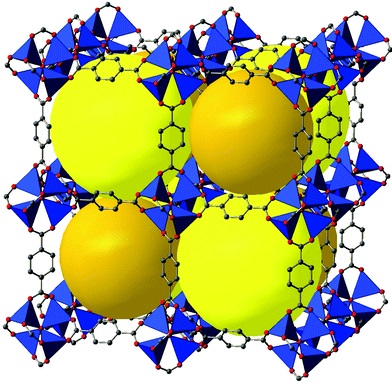
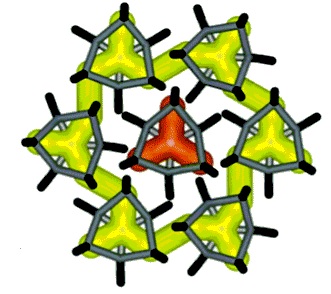
MOFs are an extremely diverse set of nanoporous materials allow for a rich set of computational studies. As a broad overview, a section of our group investigates the adsorption and kinetic separation of various gases with these materials.
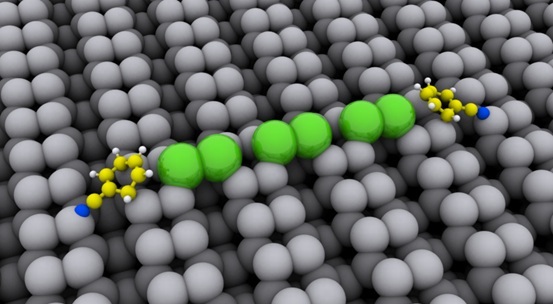
We have group members collaborating with experimentalists to develop a fundamental understanding of acid gas interactions with various MOFs, hopefully leading to the rational design of acid gas-tolerant materials with improved catalytic and separation properties.
We also perform high quality computational screening of MOFs for contaminant removal, by applying DFT methods to improve the quality of MOF data and through the calculation of high accuracy atomic point charges for charged contaminants.
In parallel with our work on zeolites, we have developed transferable ab initio based force fields, with both cluster and periodic models, for noble gas adsorption and diffusion in MOFs such as UiO-66, ZIF-8, and HKUST-1 (CuBTC).
In line with commitment to providing industrial solutions, we also assist in finding MOFs suitable for the upgrading of effluent streams from the oxidative coupling of methane (OMC) process.